Time to read: 8 min
Medical device materials must meet strict requirements for biocompatibility, sterilization compatibility, traceability, and regulatory documentation. Engineers selecting materials for medical device development must balance mechanical performance, manufacturability, cost, and regulatory compliance across the entire development lifecycle—from early prototypes to full-scale production.
Medical devices range from simple disposables to complex multi-material systems. This guide explains common materials used in medical devices, including medical-grade plastics and implantable metals, and outlines how to choose materials that meet clinical, manufacturing, and regulatory requirements.
What Are Medical-Grade Materials?
Medical-grade materials meet specific standards for use in medical devices. These standards ensure the materials are safe for contact with the human body and compatible with clinical environments.
Medical-grade materials typically meet requirements related to biocompatibility, sterilization resistance, chemical resistance, mechanical performance, regulatory documentation, and material traceability.
Importantly, a material itself is not automatically “medical grade.” The exact formulation, processing method, and supplier documentation determine whether the material qualifies for medical use. The same base polymer, for example, may exist in both industrial and medical-certified grades.
Medical device materials typically fall into three major categories: engineering plastics, implantable metals, and specialized polymers designed for sterilization and biocompatibility.
Medical Grade Plastics vs Standard Plastics
One of the most common questions in medical device development concerns the difference between medical-grade plastics and standard industrial plastics. While they may share similar base polymers, medical-grade materials meet additional requirements related to safety, traceability, and regulatory documentation.
Medical-grade plastics are produced under stricter manufacturing controls and come with certifications that support regulatory submissions. Material costs can vary significantly depending on certification level, sterilization requirements, and manufacturing volume.
| Feature | Medical-Grade Plastics | Standard Industrial Plastics |
| Regulatory documentation | Full traceability and certification | Limited documentation |
| Biocompatibility data | Often tested to ISO 10993 or USP Class VI | Usually not tested |
| Manufacturing controls | Produced under tightly controlled processes | Standard industrial manufacturing |
| Additives and colorants | Strictly controlled and documented | May vary by supplier |
| Typical applications | Medical devices, implants, surgical tools | Consumer products, industrial parts |
| Cost | Higher due to certification and traceability | Lower |
In early design stages, engineers often use industrial-grade materials for rapid prototyping. However, once devices move toward verification, validation, or clinical use, certified medical-grade materials become essential. Selecting the right material grade in the prototyping phase or early in development helps avoid costly redesigns and repeated biocompatibility testing later.
Biocompatible Materials for Medical Devices
Biocompatibility refers to a material’s ability to perform while in contact with the body without causing harmful biological reactions. The required level of testing depends on several factors, including duration of body contact, the type of tissue involved, whether the device is implanted, and whether it comes into contact with blood or bodily fluids.
ISO 10993 Biocompatibility Standards
Most regulatory pathways rely on ISO 10993, the international standard for biological evaluation of medical devices.
Common tests include:
- Cytotoxicity
- Sensitization
- Irritation
- Systemic toxicity
- Hemocompatibility
- Implantation testing
Biocompatibility testing covers more than the raw material itself. It must also account for manufacturing processes, surface treatments, cleaning procedures, sterilization methods, and any additives or colorants in the material formulation. This means a device made from a previously tested material may still require additional testing depending on how it is manufactured.
USP Class VI Materials and Testing
Another widely referenced standard is USP Class VI, which evaluates plastic materials used in medical applications through three in vivo biological reactivity tests:
Acute Systemic Toxicity Test: Evaluates systemic reactions after exposure to the material.
Intracutaneous Test: Assesses localized irritation after injection into the dermal tissue of test animals.
Implantation Test: Measures tissue response following intramuscular implantation of the material over several days.
While USP Class VI is a useful screening tool, ISO 10993 is the primary regulatory standard for medical device approval.
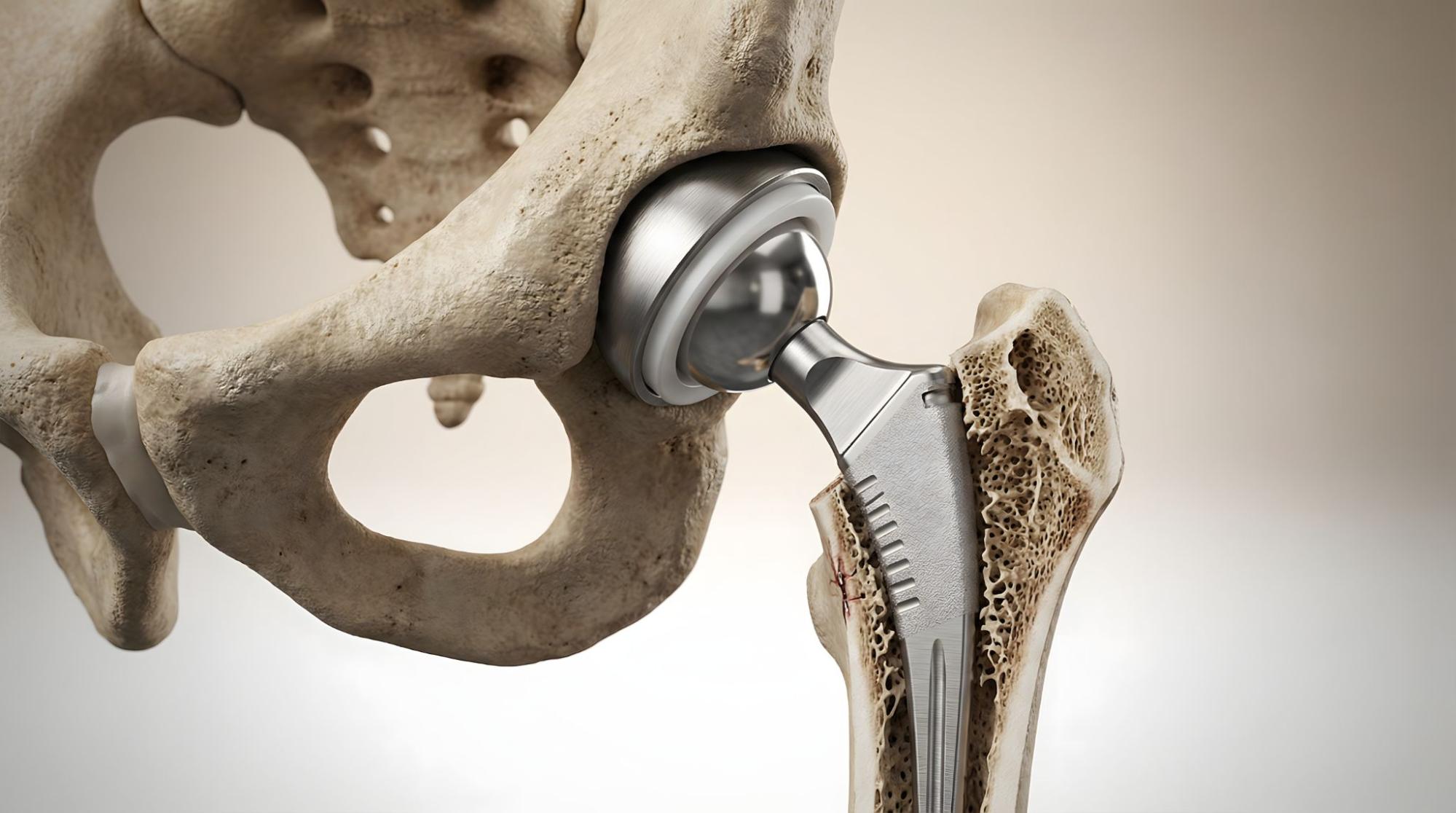
Most Commonly Used Materials in Medical Devices
Some of the most widely used materials in medical device manufacturing include polypropylene, polycarbonate, PEEK, PPSU, stainless steel 316L, titanium alloys, and cobalt-chrome. These materials are popular because they combine strong mechanical performance, sterilization compatibility, and a long history of regulatory acceptance. Medical device materials must also provide strong resistance to chemicals such as disinfectants, cleaning agents, and bodily fluids.
Plastics are often the preferred choice for disposable components, housings, and fluid-handling systems, while metals are more commonly used in structural components, surgical tools, and implants. The sections below examine commonly used plastics and metals in greater detail.
Common Medical-Grade Plastics
Plastics are widely used in medical devices for their design flexibility, chemical resistance, and cost efficiency in high-volume production. Common applications include diagnostic equipment housings, surgical instruments, drug delivery devices, catheters and tubing, disposable components, and laboratory equipment.
Polypropylene (PP)
Polypropylene is a go-to choice for disposable medical products due to its chemical resistance and low cost.
- Key properties: Chemical resistance, fatigue resistance, low cost, lightweight
- Common uses: Syringes, specimen containers, IV components, disposable medical products, packaging
- Sterilization compatibility: Compatible with ethylene oxide (EtO) and gamma radiation; limited steam sterilization tolerance
- Manufacturing compatibility: Injection molding, thermoforming
Polycarbonate (PC)
Polycarbonate is a transparent engineering plastic valued for impact resistance and optical clarity.
- Key properties: High impact strength, optical transparency, dimensional stability
- Common uses: Diagnostic equipment housings, fluid reservoirs, medical enclosures, optical components
- Sterilization compatibility: Compatible with EtO and gamma radiation; limited compatibility with repeated autoclave cycles
- Manufacturing compatibility: Injection molding, CNC machining
Polyether Ether Ketone (PEEK)
PEEK is a high-performance polymer often used in demanding medical applications.
- Key properties: Exceptional strength and stiffness, high temperature resistance, excellent biocompatibility, radiolucent
- Common uses: Orthopedic implants, spinal devices, surgical instruments, structural components
- Sterilization compatibility: Compatible with autoclave, EtO, and gamma radiation sterilization
- Manufacturing compatibility: CNC machining, injection molding, additive manufacturing
Polyphenylsulfone (PPSU)
PPSU is the material of choice for devices that need to survive repeated sterilization cycles.
- Key properties: High toughness, excellent hydrolysis resistance, long-term durability
- Common uses: Surgical instruments, sterilization trays, dental equipment, reusable medical components
- Sterilization compatibility: Excellent resistance to repeated steam autoclave cycles; also compatible with EtO and gamma sterilization
- Manufacturing compatibility: Injection molding, machining
Acetal (POM / Delrin®)
Acetal offers excellent dimensional stability and wear resistance.
- Key properties: Low friction, high stiffness, excellent dimensional stability, wear resistance
- Common uses: Device mechanisms, pump components, precision housings, moving assemblies
- Sterilization compatibility: Compatible with EtO sterilization; limited compatibility with steam and radiation sterilization
- Manufacturing compatibility: CNC machining, injection molding
Metals Used in Medical Devices
For devices that require high strength, durability, or long-term implantation, metals remain the standard choice.
Stainless Steel 316L
316L stainless steel is one of the most common metals in medical device manufacturing.
- Key properties: Corrosion resistant, high strength, durable, widely available
- Common uses: Surgical instruments, bone plates, orthopedic hardware, device structural components
- Sterilization compatibility: Compatible with steam autoclave, EtO, and radiation sterilization
- Manufacturing compatibility: CNC machining, metal forming, additive manufacturing
Titanium (Ti-6Al-4V)
Titanium alloys are often selected for implantable medical devices.
- Key properties: Exceptional strength-to-weight ratio, corrosion resistance, excellent biocompatibility
- Common uses: Dental implants, bone screws, orthopedic implants, surgical tools
- Sterilization compatibility: Compatible with steam autoclave, EtO, and radiation sterilization
- Manufacturing compatibility: CNC machining, additive manufacturing
Cobalt-Chrome (CoCr)
Cobalt-chrome alloys offer superior wear resistance.
- Key properties: Very high hardness, exceptional wear resistance, long fatigue life
- Common uses: Joint replacements, orthopedic implants, dental prosthetics
- Sterilization compatibility: Compatible with steam autoclave, EtO, and radiation sterilization
- Manufacturing compatibility: CNC machining, casting, additive manufacturing
Explore Materials.AI to discover the best materials for your next medical device design.
Comparison of Common Medical Device Materials
Medical devices use a range of materials depending on the application, mechanical requirements, and sterilization method. This table summarizes several commonly used medical device manufacturing materials.
| Material | Material Type | Key Properties | Medical Applications |
| Polypropylene (PP) | Thermoplastic | Chemical resistance, low cost | Syringes, containers, disposable devices |
| Polycarbonate (PC) | Thermoplastic | Transparency, impact resistance | Diagnostic housings, fluid reservoirs |
| Acetal (POM / Delrin) | Engineering plastic | Low friction, dimensional stability | Device mechanisms, pump components |
| PEEK | High-performance polymer | High strength, excellent biocompatibility | Orthopedic implants, surgical tools |
| PPSU | High-performance polymer | Withstands repeated sterilization | Surgical instruments, dental devices |
| Stainless Steel 316L | Metal | Corrosion resistance, strength | Surgical tools, bone plates |
| Titanium Ti-6Al-4V | Metal | Lightweight, highly biocompatible | Orthopedic implants, dental implants |
| Cobalt-Chrome | Metal | Exceptional wear resistance | Joint replacement implants |
Sterilization Requirements for Medical Device Plastics
As many devices must be sterilized before use, sterilizable plastics are an essential category of medical materials. Engineers must ensure that polymers maintain their structural integrity and performance throughout the sterilization process.
Common Sterilization Methods
Medical devices are sterilized using several methods depending on the material, device design, and manufacturing volume. Each sterilization process places thermal, chemical, or radiation stresses on device materials, which must be considered during material selection.
Steam Sterilization (Autoclave)
Steam sterilization uses high temperature and pressure to eliminate microorganisms. It’s commonly used for reusable surgical instruments and devices that must withstand repeated sterilization cycles.
Compatible materials include: Stainless steel, titanium, PPSU, and PEEK.
Ethylene Oxide (EtO)
EtO sterilization is a low-temperature chemical process used for heat-sensitive plastics and complex medical devices.
Compatible materials include: Polycarbonate, polypropylene, ABS, nylon.
Gamma Radiation
Gamma radiation sterilization is a popular choice for high-volume disposable medical devices because it can penetrate packaging and sterilize products after final assembly. However, radiation can cause embrittlement or discoloration in some polymers over time.
Compatible materials include: Polypropylene, polycarbonate, and certain medical-grade resins.
Sterilization Compatibility Comparison for Common Plastics
| Material | Autoclave | EtO | Gamma Radiation | Common Uses |
| Polypropylene | Limited | Yes | Yes | Disposable medical components |
| Polycarbonate | Limited | Yes | Yes | Diagnostic housings |
| PEEK | Yes | Yes | Yes | Implants, surgical tools |
| PPSU | Excellent | Yes | Yes | Reusable surgical instruments |
| Nylon | Limited | Yes | Limited | Mechanisms and device components |
| ABS | No | Yes | Yes | Enclosures and housings |
Selecting materials compatible with the intended sterilization method early in the design process helps prevent material degradation or device failure later.
Manufacturing Processes for Medical Device Materials
Process compatibility is critical in material selection. What works with injection molding, for example, may not be compatible with CNC machining, and vice versa.
Injection Molding
Injection molding is a standard choice for high-volume medical plastic components.
Advantages:
- Low cost per unit at scale
- Excellent repeatability
- Suitable for cleanroom manufacturing
Common materials:
- Polypropylene, polycarbonate, ABS, nylon, PEEK
CNC Machining
CNC machining supports both prototyping and production for metal and engineering plastic components.
Advantages:
- Tight tolerances
- High material variety
- Suitable for low to mid volumes
Common materials:
- PEEK, acetal (POM), stainless steel, titanium
Additive Manufacturing (3D Printing)
Additive manufacturing enables fabrication of intricate geometries and patient-specific devices.
Common uses:
- Surgical guides
- Anatomical models
- Custom implants
- Low-volume production
Common materials:
- Medical-grade nylon (PA), TPU, biocompatible photopolymers, ABS-M30i, PC-ISO
Note: Regulatory documentation, process validation, and material traceability must be carefully managed when using additive manufacturing for regulated medical devices.
Regulatory Considerations for Medical Device Materials
Medical device materials must comply with regulatory frameworks, including:
FDA (U.S. Food and Drug Administration)
Medical devices are regulated under the FDA’s Quality System Regulation (21 CFR Part 820).
Material considerations include:
- Biocompatibility documentation
- Material traceability
- Supplier qualification
- Process validation
EU MDR (European Union Medical Device Regulation)
EU MDR requires detailed documentation of:
- Material composition
- Biological safety
- Risk assessment
- Manufacturing controls
Supply Chain and Traceability
For commercial medical device manufacturing, materials must be fully traceable.
Key documentation includes:
- Certificates of analysis
- Material lot numbers
- Supplier validation
- Process records
Traceability ensures that manufacturers can identify and address potential issues during recalls or regulatory audits.
Key Considerations When Selecting Scalable Medical Device Materials
Engineers should evaluate materials according to these factors:
Biocompatibility: Does the material meet ISO 10993 or USP Class VI requirements?
Sterilization Compatibility: Can the material withstand the required sterilization method?
Manufacturability: Is the material compatible with the intended manufacturing process?
Regulatory Documentation: Is the material available with the necessary certifications?
Supply Chain Stability: Will the material remain available throughout the product lifecycle?
Read the case study to see how Fictiv helped TransMed7 accelerate product launch for five new medical devices.
Partner With Fictiv for Medical Device Manufacturing
Material selection is one of the most foundational decisions in medical device design and manufacturing. The right material must balance biological safety, mechanical performance, sterilization compatibility, manufacturability, and regulatory compliance while supporting a scalable path from prototype to production.
Fictiv helps engineering teams accelerate this process with access to a global manufacturing network, certified materials, and intelligent design tools. Using Materials.AI, engineers can quickly explore and compare material options based on performance requirements, helping identify viable candidates faster and reducing development risk. By combining AI-powered material insights with production-ready manufacturing services, Fictiv enables teams to move more efficiently from early prototypes to validated production parts.
Get started with a free quoteFAQs About Medical Device Materials
What materials are commonly used in medical devices?
Common plastics include polypropylene, polycarbonate, PEEK, PPSU, nylon, and acetal. Metals such as stainless steel 316L, titanium alloys, and cobalt-chrome are widely used due to their strength, corrosion resistance, and biocompatibility.
What makes a material medical grade?
Medical-grade materials meet strict standards for biocompatibility, traceability, manufacturing control, and regulatory documentation.
What is ISO 10993?
ISO 10993 is the international standard used to evaluate the biocompatibility of medical device materials and their interaction with biological systems.
How does Fictiv support medical device material selection?
Fictiv helps engineering teams select medical device materials by providing access to vetted suppliers, certified materials, and data-driven tools like Materials.AI. Engineers can compare material properties, evaluate manufacturability, and ensure compatibility with sterilization and regulatory requirements early in development.